Simulations of chemical reactions
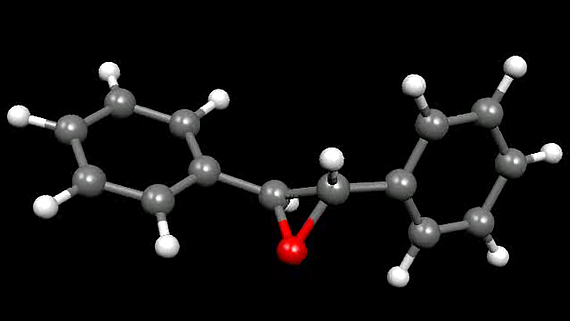
The evolution of quantum chemistry has got to a point at which it is possible not only to compute molecular structures, but also to simulate chemical reactions on the basis of the elementary quantum mechanical equations. We investigate ultrafast chemical reactions using first principles molecular dynamics simulations with the Car-Parrinello program package (using the celebrated Car-Parrinello concept: description of the chemically relevant valence electrons with density functional theory in the Kohn-Sham formalism with a plane wave basis set, description of the core electrons with fixed potentials, description of the motion of the nuclei with classical Newton dynamics).
From the simulation the full information about the mechanism is obtained (motion of nuclei and electronic orbitals, entropy, reaction time). However, the time scale of molecular motion is the femtosecond time scale. To fully model the molecular vibrations with periods of some ten femtoseconds, very small time steps have to be used in the simulations and total simulation times presently hardly excess the picosecond time scale. If watched at this scale in extreme slow motion (and extreme magnification), molecules turn out to be rather inactive and chemical reactions appear as very rare events. Even an ultrafast decomposition looks like a slow random walk in the simulation. From time to time molecules collide, eventually exchange electrons or protons, bonds may be formed or broken. Hence, in order not to observe just unreactive molecular oscillations (which could also be modelled with much cheaper computational approaches), we focus on highly reactive chemical systems.
We use the following approaches for preparing highly reactive systems:
- Switching to the potential surface of an excited state (photochemistry)
- Expanding or compressing a molecular system (mechanically induced chemistry)
- Mixing of liquid or gaseous components (chemistry)
- Adding or removing electrons (electrochemistry)
- Applying external fields (e.g. electrochemistry)
- Bringing compounds in contact with a reactive surface (surface chemistry)
Photoreactions
Theoretical description of photoreactions is particularly challenging: since calculating electronically excited states is usually more demanding than calculating the ground states and since the reaction coordinates are often unknown.
Using the ROKS method (Restricted Open-shell Kohn-Sham), we perform molecular dynamics in the first excited singlet state.[7, 8, 9] There, the atomic nuclei move (according to the laws of classical mechanics) in an electronic potential which is calculated using quantum chemical methods. With this approach we investigated for example photoreactions of small, organic molecules (butadiene, cyclohexadiene), as well as the photoreaction of azobenzene, a molecular rotor and of rhodopsin (the latter in combination with QM/MM).
Movie 1 shows the photoreaction of oxirane: immediately after excitation the C-O bond breaks in agreement with experimental results, subsequent hydrogen migration leads to an aldehyde. A large amount of the released energy is transfered to the vibrational degrees of freedom.
Photochemical excitation of diphenyloxirane leads to cleavage of the C-C bond in a complicated disrotatory motion, as shown in movie 2.
Movie 1 [oxiran]
Movie 2 [diphenyl-lang]
An other reaction type induced by electronic excitation is the photodissociation. Movie 3 shows the cleavage of the N-H bond in the pyrrole molecule. The anti-binding orbital of the excited state is converted into a spherical hydrogen orbital.
The reaction is modified in the presence of solvents. The dissociation is prevented in a pyrrole•3H2O cluster (movie 4). Instead an electron transfer to the water molecules is observed (orange orbital). Movie 5 shows a pyrrole•3NH3 cluster. In this more basic system the electron transfer is followed by the transfer of the proton.
Movie 3 [lumo]
Movie 4 [pyrrol_PW3]
Movie 5 [pyrrol_3NH3]
Reactions under mechanical stress
We investigated the material failure of siloxane materials under mechanical load in cooperation with the Wacker Chemie AG. In contrast to previous models which are based on radical bond rupture, the Car-Parrinello molecular dynamics simulations show an ionic bond rupture. This observation is in accordance with ESR-experiments. Hence the Si-O and C-C bonds exhibit a qualitatively different rupture behaviour while they have comparable bond strenghts. This affects the consecutive reactions: the ionic fragments react quickly with neighbouring chains. Due to this mechanism a crack propagates rapidly.
Movie 1 shows the tearing of a siloxane chain from a silica surface. In movie 2, four siloxane chains under mechanical stress are shown.
Motivated by single molecule AFM experiments, we are investigating chemical reactions induced by pulling a disulfide with first principles simulations. Both in the presence (reduction) and in the absence (simple bond rupture) of the reducing agent dithiothreitol, we investigate the force to break the disulfide bond.
The relevant localized orbitals are depicted in short movies. Movie 3 shows the disulfide in the absence of solvent and reducing agent, movie 4 shows the disulfide in ammonia and in the presence of dithiothreitol.
Movie 1 [vert50]
Movie 2 [cond]
Movie 3 [disulfid]
Movie 4 [system]
Highly reactive mixtures
Hypergolic mixtures are compounds that react spontaneously when brought into contact with one another. This characteristic is deployed e.g. in the propellant for the upper stage engine of the European launch vehicle Ariane 5. The bipropellant that is used in this case consists of monomethylhydrazine (MMH) and nitrogen tetroxide (NTO). Complications in the upper stage procedure emerged during a mission to transport two satellites into space in July 2001. A possible reason for this could be undesired reactions in the bipropellant.
We investigate the reactions of the MMH/NTO mixture on a picosecond timescale with Car-Parrinello molecular dynamics.
We observe the sequence of electron (MMH→NTO) and proton transfers (e.g. MMH radical cation→MMH) over a few picoseconds. The proton transfers are shown in movies 1 and 2 which present a timeframe of two picoseconds. The first electron transfer of this sequence is shown in movie 3.
Movie 1 [mmh1]
Movie 2 [mmh2]
Movie 3 [mmhorb]
Surface chemistry
The movie shows the reaction of liquid air with aluminium. The oxygen molecules (red) react readily with the surface while the nitrogen molecules remain inactive. The reaction is known as being highly exothermic and a large amount of energy is released each time an oxygen molecule reacts with the surface. The oxygen molecules separate into atoms which stick strongly to the surface at an average distance of roughly 4 Angstrom. Finally also the one water molecule contained in the simulation box reacts with a surface oxygen atom to yield two hydroxyl groups. At that point, the surface is already close to melting, as the energy is not removed from the simulation box. While the oxygen atoms are strongly bonded to the surrounding aluminium atoms, the underlying metal surface is ductile and allows for some mobility of the surface aluminium oxide islands.
Movie [aluminium]
Electrochemistry
The electrolysis of water is a prime example of an electrolytic reaction. Water is decomposed into H2 and O2 by an electric current between two electrodes. Understanding the reaction mechanism at the atomic level gives insight into the basic concepts of electrochemical reactivity. With Car-Parrinello molecular dynamics simulations the elementary reaction steps of the electrolysis of bulk water are investigated. We focus on the reactions in pure water without explicit consideration of the electrode surfaces. Our simulations illustrate the single reaction steps starting from the discharged solutions to the formation of the reaction products.
Movie 1 (the evolution of triplet molecular oxygen. Depicted are the spin densities) shows a simulation starting from eight OH• radicals (four with spin up and four with spin down, as is evident from the spin densities shown). At the end of the movie the aqueous solution contains one triplet oxygen molecule (O2), two OH• radicals, and one hydrogen peroxide molecule (H2O2) in addition to hydroxide anions and hydronium cations.
Movie 2 ( the evolution of molecular hydrogen. The orbitals shown are the localized (Wannier) orbitals of the excess electrons) shows the formation of one hydrogen molecule (H2) from two H3O+ cations and two excess electrons.
Movie 1 [o2-evolution]
Movie 2 [H2-DerOrbitalfilm]
External fields
We have implemented a sinusoidal potential which may change in phase and amplitude. In first applications electron transfer and ion migration are modeled.
Electron transfer in a single molecule junction in an external field.
Movie 1: Motion of the electrons shown as centers of localized orbitals.
Movie 2: Motion of a single orbital.
Movie 1 [name_film]
Movie 2 [ORB-296]
Contact


30167 Hannover




